Products
Featured products
VHP Pass Box Manufacturer | 6-Log Sterile Transfer Solution
Boben VHP Vaporized Hydrogen Peroxide Pass-Through Chamber: Achieving 6-Log Sterile Transfer
Employing vaporized hydrogen peroxide (VHP) sterilization technology compliant with international GMP standards, the Boben pass-through system achieves a 99.9999% (6-Log) spore kill rate. Specifically engineered for pharmaceutical, biotechnology, and high-biosafety-level laboratories, this equipment vaporizes H₂O₂ into a high-concentration sterilizing gas. This process delivers exceptional penetration power, ensuring every nook and cranny within the transfer chamber achieves a sterile state.
Features:
- VHP sterilization achieves 99.999% effectiveness with no residue
- 304 stainless steel + double-sealed construction prevents cross-contamination
- Interlocking design prevents accidental operation for enhanced safety
- One-touch control with adjustable parameters for diverse materials
- Universal voltage + multilingual interface for global use
- SGS/CE certified for compliance and easy approval
- Durable core components with 1-year warranty + worldwide after-sales support
Product Overview
Our VHP Pass Box employs a vaporized hydrogen peroxide (VHP) transfer system compliant with GMP standards, recognized internationally as an efficient sterile transfer solution. Compared to traditional UV and ozone disinfection, our Vaporized H₂O₂ technology offers significant advantages including superior penetration, rapid sterilization, and no residuals.
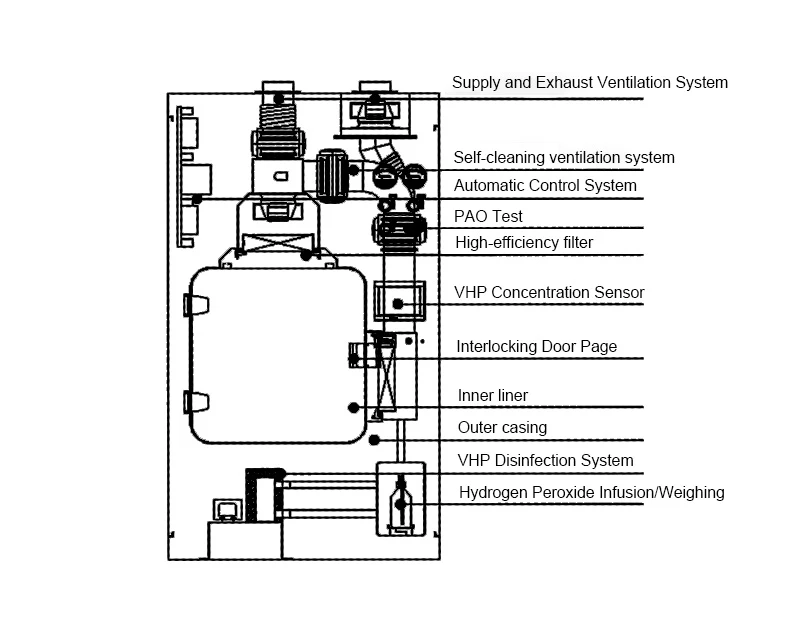
By vaporizing hydrogen peroxide solution into a high-concentration sterilizing gas, the system rapidly permeates every corner of the transfer chamber, including crevices and dead zones. It achieves a 99.9999% (6-Log) inactivation rate against bacteria, viruses, and spores, fully complying with international standards such as ISO 14644 and GMP. This makes it the core barrier for achieving Grade A Sterile Transfer.
Product Information
| Model | Internal Dimensions (W*D*H) | External Dimensions (W*D*H) | Power (kw) | Weight (kg) |
|---|---|---|---|---|
| KD-111 | 600*600*600mm | 1050*650*1800mm | 2 | 280 |
| KD-112 | 600*600*800mm | 1050*650*2000mm | 2.2 | 300 |
| KD-113 | 600*1000*600mm | 1050*1050*1800mm | 2.2 | 320 |
| KD-114 | 800*800*800mm | 1250*850*2000mm | 2.3 | 350 |
| KD-115 | 800*800*1000mm | 1250*850*2200mm | 2.5 | 360 |
| KD-116 | 800*1000*1000mm | 1250*1050*2200mm | 2.6 | 380 |
| KD-117 | 800*1000*1200mm | 1250*1050*2200mm | 2.8 | 400 |
| KD-118 | 800*1200*1600mm | 1450*1250*2150mm | 3 | 450 |
| KD-119 | 800*1400*1600mm | 1450*1450*2150mm | 3.2 | 480 |
| KD-120 | 1200*1400*1600mm | 1850*1450*2150mm | 4 | 600 |
Core Technology Advantages
- 6-Log Sterilization Efficiency: Guaranteed bio-decontamination using high-concentration VHP.
- Siemens PLC Control System: Fully automated cycle management with a user-friendly HMI touch screen.
- Inflatable Gasket Sealing: High-grade Inflatable Seals ensure a leak-free environment for maximum safety.
- SS316L Internal Chamber: Superior corrosion resistance against H2O2, built with medical-grade stainless steel.
- Integrated VHP Generator: A compact, all-in-one system designed for space-saving and rapid setup.
- Electromagnetic Interlock: Prevents cross-contamination by ensuring dual doors never open simultaneously.
Technical Specifications Table
| Feature | Specification Details |
| Material | Internal SS316L / External SS304 |
| Sterilization Rate | 6-Log Reduction (Geobacillus stearothermophilus) |
| Control System | Siemens PLC with Data Logging & Audit Trail |
| Seal Type | Inflatable Gasket (Compressed Air) |
| Cycle Time | 30 – 55 Minutes (Adjustable Aeration) |
| Compliance | GMP, ISO 14644, CE Certified |
VHP Pass Box Application Scenarios
- Cell & Gene Therapy: Safe transfer of sensitive biological materials.
- BSL-3 & BSL-4 Laboratories: High-containment bio-safety transfer solutions.
- Pharmaceutical Manufacturing: Grade A aseptic area material air locks.
- Modular Cleanroom Interfaces: Seamless integration into existing cleanroom walls.
VHP Pass Box Detailed Display
1.VHP Pass Box with High-Quality Stainless Steel Outer Casing
The outer chamber is constructed from premium, thickened stainless steel (such as SUS304 or 316L). This reinforced structure not only withstands physical impacts during daily operation but also ensures the chamber maintains excellent flatness and sealing integrity without deformation under high-frequency pressure fluctuations. The VHP sterilization process imposes stringent oxidation requirements on equipment materials. Our stainless steel surfaces undergo specialized surface treatment, delivering exceptional corrosion resistance. Even with prolonged exposure to highly oxidizing sterilizing steam, it effectively prevents rust and pitting corrosion, ensuring extended equipment lifespan. The outer casing features a smooth, seamless finish with rounded corners and welded joints, eliminating potential bacterial breeding sites. This easy-to-clean design simplifies daily maintenance—a simple wipe restores a highly sanitary state, fully meeting GMP hygiene and safety requirements.

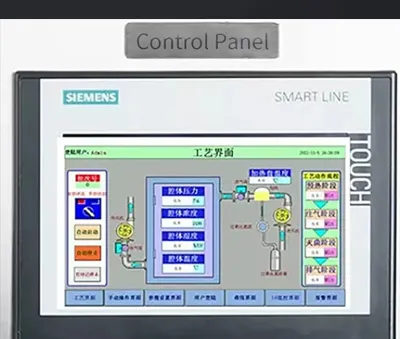
2.VHP Pass Box Intelligent PLC Control System Analysis
Equipped with a Siemens SMART LINE dedicated control panel, the process flow is clearly visible at a glance. Users can monitor core data such as chamber pressure, sterilization concentration, and remaining time in real-time via the full-color touchscreen, eliminating the lag and ambiguity of traditional mechanical gauges. Built-in professional variable-frequency logic algorithms precisely adjust ventilation volume and sterilization intensity according to different process requirements. This not only ensures uniform distribution of VHP gas but also effectively extends the service life of core components, achieving a dual balance of efficiency and energy savings. The control panel integrates an emergency power button (E-Stop) to instantly halt operation in extreme abnormal situations, safeguarding personnel and materials. Additionally, the system supports multi-level user permission management to prevent sterilization program interruptions caused by misoperation.
3.VHP Pass Box High-Precision Differential Pressure Gauge Analysis
This differential pressure gauge employs a classic mechanical structure, requiring no external power source for operation. This ensures 24/7 uninterrupted real-time monitoring even in complex laboratory environments or during unexpected power outages, guaranteeing the safe continuity of critical processes.Exceptional Sensitivity and Response Speed: The gauge demonstrates outstanding sensitivity to minute pressure fluctuations during VHP sterilization. It detects even the slightest changes in chamber air pressure, providing instant feedback via a responsive pointer. This enables operators to instantly verify seal integrity and sterilization environment stability.Industrial-Grade Measurement Accuracy: Each differential pressure gauge undergoes precise calibration, delivering exceptional measurement accuracy. Its clear, easy-to-read dial design facilitates rapid data recording by technicians, fully meeting the stringent audit requirements for differential pressure monitoring in the biopharmaceutical and precision manufacturing industries.


4.VHP Pass Box High-Efficiency Filtration System Analysis
The filter mesh employs advanced monolithic molding technology, featuring an aesthetically pleasing design with a tightly sealed structure. This integrated construction not only enhances the equipment’s industrial aesthetics but, more importantly, eliminates potential frame leakage risks associated with traditional assembled filters, ensuring every inch of airflow undergoes rigorous purification. The core filter element delivers exceptional filtration precision, effectively capturing airborne particulates, bacteria, and microorganisms. Combined with stable airflow organization, it rapidly achieves an extremely high cleanliness level inside the pass box, meeting the most stringent production standards. Safety during dynamic operation was thoroughly considered from the system’s inception. Through continuous high-efficiency filtration, it effectively prevents the introduction of external contaminants during material transfer, completely eliminating secondary contamination in the workshop and safeguarding your GMP-compliant production.
As a manufacturer specializing in cleanroom technology, we understand the stringent standards of the Middle East and global markets. Whether you’re seeking sandwich panel suppliers in the UAE or laboratory equipment in Saudi Arabia, we offer ISO-certified solutions directly from our factory.Contact us now or add WhatsApp +86 138 6252 7051 to receive a professional quote and technical support tailored to your project.
Validation & Support
Your compliance is our top priority.We understand that documentation is as critical as the equipment itself. Boben provides comprehensive IQ/OQ/PQ validation solutions to ensure your facility passes any regulatory audit. Whether you’re in Saudi Arabia, Turkey, or Australia, our engineering team delivers global on-site technical support.
Case Studies
FAQ
A: Yes, we specialize in custom Stainless Steel VHP Chambers tailored to your specific cleanroom layout.
Need the VHP Pass Box Price for your next project? Contact us for a free 3D design and technical consultation.
Boben Factory Environment
Add us on WhatsApp to learn more about Boben and get a free design proposal.
Act Now:Customize Your Rapid Clean Space!
Don’t let lengthy lead times slow down your production schedule.Contact our cleanroom experts for a free design consultation and customized quote to kickstart your fast-track production journey.































